 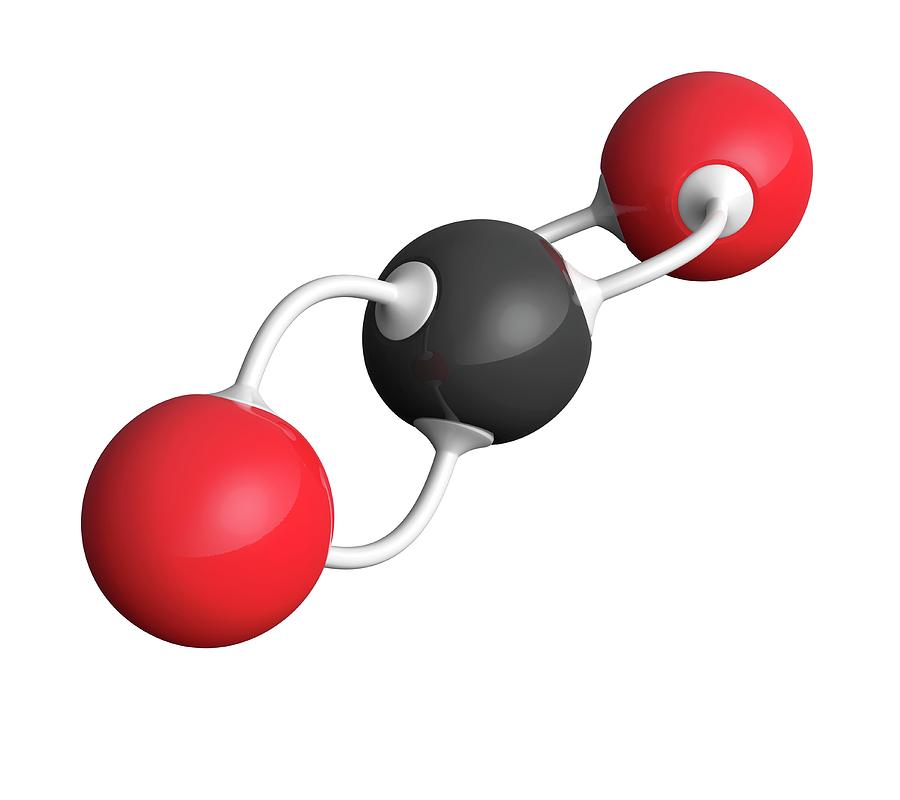
Carbon dioxide is the most significant GHG. The accumulation of gases released from fossil fuel incineration in the atmosphere results in the trapping of heat within the earth, which is technically referred to as the greenhouse gas (GHG) effect. More information about the utilization and economy of carbon dioxide could be found elsewhere. The utilization of carbon dioxide as a reactant in manufacturing chemicals also helps reduce the aftereffects of its emission. Therefore, carbon dioxide capture and reuse are attracting significant attention. The concentration of carbon dioxide is predicted to reach about 26 billion tons/year in 2100. The increasing consumption of hydrocarbons has induced a 30% increase in the concentration of carbon dioxide since the preindustrial period. It is this greenhouse warming that makes life, as we know it, possible on planet Earth. water vapor, methane, nitrous oxide, and chlorofluorocarbons) helps keep the planet warmer than it would be without an atmosphere. 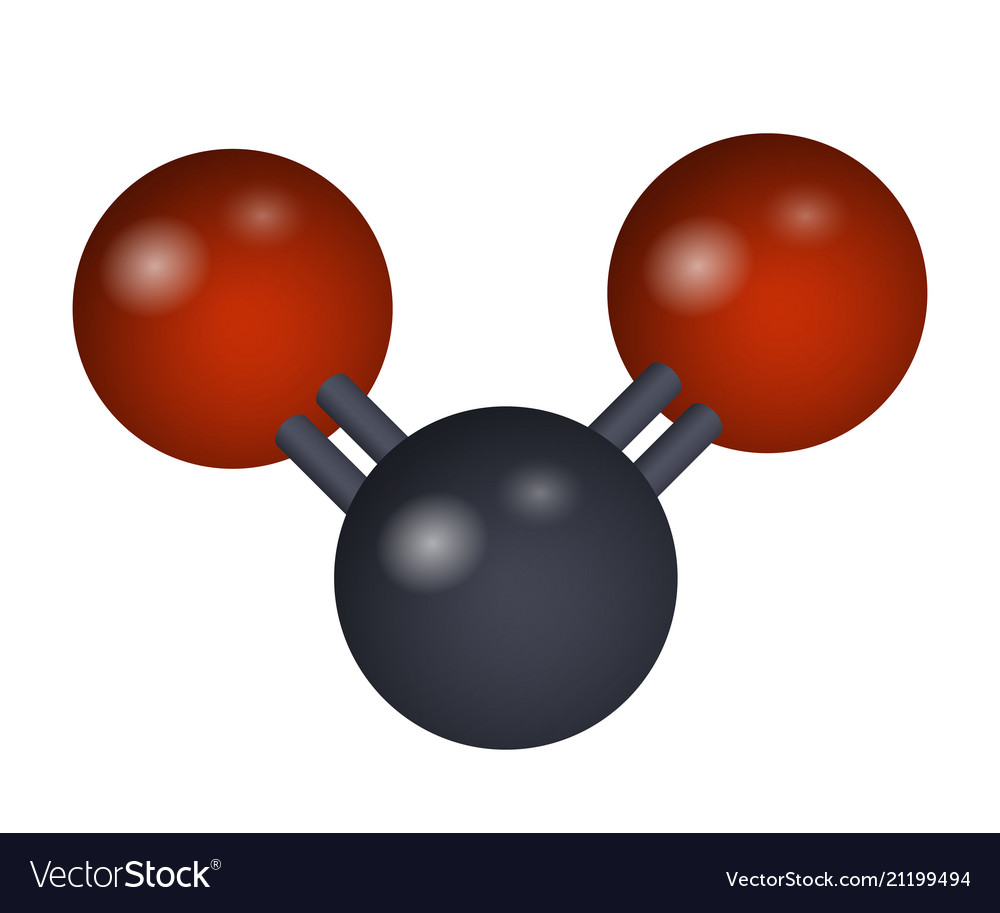
The trapping of radiation by CO 2 and other greenhouse gases (e.g. CO 2 is also a greenhouse gas that absorbs long-wavelength radiation in the atmosphere, attenuating its escape into space. For example, atmospheric CO 2 concentrations in the Northern Hemisphere are generally lower in the summer, when many plant species enter a new growth stage and photosynthesis predominates over respiration. Variations in the global balance between photosynthesis and respiration result in seasonal variations in atmospheric CO 2 of up to 15 ppm. Plants take up CO 2 and release oxygen as a part of photosynthesis. Despite its low concentrations relative to those of nitrogen or oxygen, CO 2 plays a significant role in the Earth's life cycle and in controlling the global climate.ĬO 2 is released as a by-product of aerobic respiration. Feely, in Encyclopedia of Atmospheric Sciences, 2003 IntroductionĬarbon dioxide (CO 2) is considered a trace gas in the atmosphere, with contemporary concentrations of approximately 370 parts per million by volume (ppm). Inhalation of 6–10% causes dyspnea, headache, dizziness, sweating, and restlessness.Ĭ.L. Unconsciousness may occur when inhaling a concentration of 12% for 8–23 min. Carbon dioxide concentrations of 20–30% can cause convulsions and coma within 1 min. The lowest lethal concentration (inhalation) for humans is 100000 ppm for 1 min. Increased fetal movements have been noted in humans following inhalation with 5% carbon dioxide in air. Inhalation of carbon dioxide is teratogenic and has caused both male and female adverse reproductive effects in rodents. Cough, headache, fever, malaise, limb swelling, and unconsciousness were noted in the victims. The Lake Nyos disaster in August 1986 has been postulated to have resulted from the release of carbon dioxide from rising cold deep water producing a deadly cloud of gas. Combined respiratory and metabolic acidosis was seen in a serious exposure to dry ice. Decreased vision and increased intraocular pressure may be seen with inhalation of 10% carbon dioxide. Gastrointestinal effects, like nausea and vomiting, may occur, but usually resolve within 24–48 h following termination of exposure. Prolonged hypoxia may result in unconsciousness seizures may be seen during serious cases of asphyxia. Various neurologic effects like dizziness, headaches, sleepiness, and mental confusion can occur. Respiratory effects like hyperventilation, cyanosis, and pulmonary edema are also noted. It is also a vasodilator and the most potent cerebrovascular dilator known. Carbon dioxide exerts a direct toxic effect to the heart, resulting in diminished contractile force. Four stages have been described (depending on the arterial oxygen saturation): (1) indifferent stage, 90% oxygen saturation (2) compensatory stage, 82–90% oxygen saturation (3) disturbance stage, 64–82% oxygen saturation and (4) critical stage, 60–70% oxygen saturation or less.įollowing exposure to asphyxiants, cardiovascular effects like tachycardia, arrhythmias, and ischemia are noted. Mehendale, in Encyclopedia of Toxicology (Second Edition), 2005 HumanĬarbon dioxide is a simple asphyxiant that displaces oxygen from the breathing atmosphere resulting in hypoxia. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
